Lactato deshidroxenase: Diferenzas entre revisións
Sen resumo de edición |
Sen resumo de edición |
||
| Liña 91: | Liña 91: | ||
A lactato deshidroxenase ten importancia médica porque se encontra abundantemente nos tecidos corporais, como o músculo e células do [[sangue]]. Como se libera cando hai danos nos tecidos, serve como marcador de lesións e doenzas comúns. |
A lactato deshidroxenase ten importancia médica porque se encontra abundantemente nos tecidos corporais, como o músculo e células do [[sangue]]. Como se libera cando hai danos nos tecidos, serve como marcador de lesións e doenzas comúns. |
||
Existen catro clases de lactato deshidroxenase, que poden actuar sobre o D-lactato ([[D-lactato deshidroxenase (citocromo)]] |
Existen catro clases de lactato deshidroxenase, que poden actuar sobre o D-lactato ou o L-lactato e ser dependentes do [[citicromo c]] ou do NAD(P), que son: [[D-lactato deshidroxenase (citocromo)]], [[D-lactato deshidroxenase (NAD(P)), [[L-lactato deshidroxenase (citocromo)]] e [[L-lactato deshidroxenase (NAD(P)). Este artigo trata sobre a '''L-lactado deshidroxenase dependente de NAD(P)'''. |
||
==Reaccións== |
==Reaccións== |
||
[[Ficheiro:LDH reaction.png|left|miniatura|355px|Reacción que cataliza a LDH.]] |
[[Ficheiro:LDH reaction.png|left|miniatura|355px|Reacción que cataliza a LDH.]] |
||
A lactato deshidroxenase cataliza a interconversión reversible do [[piruvato]] e o [[lactato]] á vez que se interconverten o [[NADH]] e o [[NAD]]<sup>+</sup>. O piruvato é o produto final da [[glicólise]], que cando non hai oxíxeno ou é moi escaso, fermenta a lactato. Despois, realízase a reacción inversa durante o [[ciclo de Cori]] no [[fígado]]. A altas concentracións de lactato, o encima mostra [[inhibición encimática|inhibición]] por retroalimentación, e a velocidade de conversión do piruvato en lactato diminúe. |
|||
Lactate dehydrogenase catalyzes the interconversion of [[pyruvate]] and [[lactic acid|lactate]] with concomitant interconversion of NADH and [[Nicotinamide adenine dinucleotide|NAD<sup>+</sup>]]. It converts pyruvate, the final product of [[glycolysis]], to lactate when oxygen is absent or in short supply, and it performs the reverse reaction during the [[Cori cycle]] in the [[liver]]. At high concentrations of lactate, the enzyme exhibits feedback inhibition, and the rate of conversion of pyruvate to lactate is decreased. |
|||
Tamén pode catalizar a deshidroxenación do [[2-hidroxibutirato]], pero é un [[substrato encimático|substrato]] moito peor que o lactato para o encima. Ten moi pouca ou ningunha actividade co [[beta-hidroxibutirato]]. |
|||
It also catalyzes the dehydrogenation of [[2-Hydroxybutyrate]], but it is a much poorer substrate than lactate. There is little to no activity with [[beta-hydroxybutyrate]]. |
|||
==Regulación do encima== |
==Regulación do encima== |
||
Esta proteína pode usar o modelo da [[morfeeína]] de [[regulación alostérica]].<ref name="pmid22182754">{{cite journal | author = Selwood T, Jaffe EK | title = Dynamic dissociating homo-oligomers and the control of protein function | journal = Arch. Biochem. Biophys. | volume = 519 | issue = 2 | pages = 131–43 | year = 2012 | month = March | pmid = 22182754 | pmc = 3298769 | doi = 10.1016/j.abb.2011.11.020 }}</ref> |
|||
===Hipoglicemia inducida por etanol=== |
===Hipoglicemia inducida por etanol=== |
||
| Liña 108: | Liña 108: | ||
The increased NADH/NAD+ ratio also can cause hypoglycemia in an (otherwise) fasting individual who has been drinking and is dependent on gluconeogenesis to maintain blood glucose levels. Alanine and lactate are major gluconeogenic precursors that enter gluconeogenesis as pyruvate. The high NADH/NAD+ ratio shifts the lactate dehydrogenase equilibrium to lactate, so that less pyruvate can be formed and, therefore, gluconeogenesis is impaired. |
The increased NADH/NAD+ ratio also can cause hypoglycemia in an (otherwise) fasting individual who has been drinking and is dependent on gluconeogenesis to maintain blood glucose levels. Alanine and lactate are major gluconeogenic precursors that enter gluconeogenesis as pyruvate. The high NADH/NAD+ ratio shifts the lactate dehydrogenase equilibrium to lactate, so that less pyruvate can be formed and, therefore, gluconeogenesis is impaired. |
||
== |
== Isoencimas == |
||
A lactato deshidroxenase funcional é un homo ou heterotetrámero composto polas subunidades M e H codificads polos xenes ''LDHA'' do [[cromosoma 11]] e ''LDHB'' do [[cromosoma 12]], respectivamente. As posibles [[isoencima]]s son: |
|||
Functional lactate dehydrogenase are homo or hetero tetramers composed of M and H protein subunits encoded by the ''LDHA'' and ''LDHB'' genes, respectively: |
|||
* LDH-1 (4H) |
* LDH-1 (4H) — presente no [[corazón]] e [[glóbulo vermello|glóbulos vermellos]] do sangue |
||
* LDH-2 (3H1M) |
* LDH-2 (3H1M) — no [[sistema reticuloendotelial]] |
||
* LDH-3 (2H2M) |
* LDH-3 (2H2M) — nos [[pulmón]]s |
||
* LDH-4 (1H3M) |
* LDH-4 (1H3M) — nos [[ril]]es, [[placenta]], e [[páncreas]] |
||
* LDH-5 (4M) |
* LDH-5 (4M) — no [[fígado]] e [[músculo esquelético]]<ref>{{cite book |author=Van Eerd, J. P. F. M.; Kreutzer, E. K. J. |title=Klinische Chemie voor Analisten deel 2 |pages=138–139 |year=1996 |isbn=978-90-313-2003-5}}</ref> |
||
O principal isoencimas presentes no fígado e músculo esquelético é o M<sub>4</sub>, que ten catro subunidades M (M do inglés ''muscle''), mentres que o isoencima H<sub>4</sub> é o principal na maioría das especies no corazón, con 4 subunidades H (H de ''heart''). As outras variantes conteñen as dúas subunidades en diferentes proporcións, polo que son heterotetrámeras. |
|||
The five isoenzymes that are usually described in the literature each contain four subunits. The major isoenzymes of skeletal muscle and liver, M<sub>4</sub>, has four muscle (M) subunits, while H<sub>4</sub> is the main isoenzymes for heart muscle in most species, containing four heart (H) subunits. The other variants contain both types of subunits. |
|||
Xeralmente a LDH-2 é a forma predominante no [[soro sanguíneo]]. Un nivel de LDH-1 máis alto que o de LDH-2 (un "patrón invertido" con respecto ao normal) suxire [[infarto de miocardio]] (as lesións cardíacas liberan a LDH cardíaca LDH-1, no torrente sanguíneo). |
|||
Usually LDH-2 is the predominant form in the [[Blood serum|serum]]. A LDH-1 level higher than the LDH-2 level (a "flipped pattern") suggests [[myocardial infarction]] (damage to heart tissues releases heart LDH, which is rich in LDH-1, into the bloodstream). The use of this phenomenon to diagnose infarction has been largely superseded by the use of [[Troponin]] I or T measurement.{{Citation needed|date=October 2013}} |
|||
==Xenética en humanos== |
==Xenética en humanos== |
||
Revisión como estaba o 28 de decembro de 2013 ás 18:36
Este artigo está a ser traducido ao galego por un usuario desta Wikipedia; por favor, non o edite. O usuario Miguelferig (conversa · contribucións) realizou a última edición na páxina hai 10 anos. Se o usuario non publica a tradución nun prazo de trinta días, procederase ó seu borrado rápido. |
| Lactato deshidroxenase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identificadores | |||||||||
| Número EC | 1.1.1.27 | ||||||||
| Número CAS | 9001-60-9 | ||||||||
| Bases de datos | |||||||||
| IntEnz | vista de IntEnz | ||||||||
| BRENDA | entrada de BRENDA | ||||||||
| ExPASy | vista de NiceZyme | ||||||||
| KEGG | entrada de KEGG | ||||||||
| MetaCyc | vía metabólica | ||||||||
| PRIAM | perfil | ||||||||
| Estruturas PDB | RCSB PDB PDBe PDBj PDBsum | ||||||||
| Gene Ontology | AmiGO / EGO | ||||||||
| |||||||||

| |
| Lactato deshidroxenase M4 humana (o isoencima que se encontra no músculo esquelético). De PDB 1I10. | |
Lactato deshidroxenase
| |
| Identificadores | |
| Símbolo | LDHA |
| Símbolos alt. | LDHM |
| Entrez | 3939 |
| HUGO | 6535 |
| OMIM | |
| RefSeq | NM_005566 |
| UniProt | P00338 |
| Outros datos | |
| Número EC | 1.1.1.27 |
| Locus | Cr. 11 p15.4 |
Lactato deshidroxenase
| |
| Identificadores | |
| Símbolo | LDHB |
| Símbolos alt. | LDHL |
| Entrez | 3945 |
| HUGO | 6541 |
| OMIM | |
| RefSeq | NM_002300 |
| UniProt | P07195 |
| Outros datos | |
| Número EC | 1.1.1.27 |
| Locus | Cr. 12 p12.2-12.1 |
Lactato deshidroxenase
| |
| Identificadores | |
| Símbolo | LDHC |
| Entrez | 3948 |
| HUGO | 6544 |
| OMIM | |
| RefSeq | NM_002301 |
| UniProt | P07864 |
| Outros datos | |
| Número EC | 1.1.1.27 |
| Locus | Cr. 11 p15.5-15.3 |
| Lactato deshidroxenase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
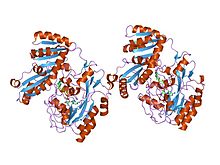 Estrutura cristalina da D-lactato deshidroxenase, un encima respiratorio de membrana periférico. | |||||||||
| Identificadores | |||||||||
| Símbolo | Lact-deh-memb | ||||||||
| Pfam | PF09330 | ||||||||
| Pfam clan | CL0277 | ||||||||
| InterPro | IPR015409 | ||||||||
| SCOPe | 1f0x / SUPFAM | ||||||||
| |||||||||
A lactato deshidroxenase (LDH ou LD) é un encima que se encontra en animais, plantas e procariotas, que intervén na fermentación homoláctica. Como deshidroxenase que é transfire un hidróxeno dunha molécula a outra. En concreto, a lactato deshidroxenase cataliza a conversiónn reversible do piruvato en lactato e á vez converte o coencima NADH na súa forma oxidada NAD+.
A lactato deshidroxenase ten importancia médica porque se encontra abundantemente nos tecidos corporais, como o músculo e células do sangue. Como se libera cando hai danos nos tecidos, serve como marcador de lesións e doenzas comúns.
Existen catro clases de lactato deshidroxenase, que poden actuar sobre o D-lactato ou o L-lactato e ser dependentes do citicromo c ou do NAD(P), que son: D-lactato deshidroxenase (citocromo), [[D-lactato deshidroxenase (NAD(P)), L-lactato deshidroxenase (citocromo) e [[L-lactato deshidroxenase (NAD(P)). Este artigo trata sobre a L-lactado deshidroxenase dependente de NAD(P).
Reaccións

A lactato deshidroxenase cataliza a interconversión reversible do piruvato e o lactato á vez que se interconverten o NADH e o NAD+. O piruvato é o produto final da glicólise, que cando non hai oxíxeno ou é moi escaso, fermenta a lactato. Despois, realízase a reacción inversa durante o ciclo de Cori no fígado. A altas concentracións de lactato, o encima mostra inhibición por retroalimentación, e a velocidade de conversión do piruvato en lactato diminúe.
Tamén pode catalizar a deshidroxenación do 2-hidroxibutirato, pero é un substrato moito peor que o lactato para o encima. Ten moi pouca ou ningunha actividade co beta-hidroxibutirato.
Regulación do encima
Esta proteína pode usar o modelo da morfeeína de regulación alostérica.[1]
Hipoglicemia inducida por etanol
Ethanol is dehydrogenated to acetaldehyde by alcohol dehydrogenase, and further into acetic acid by acetaldehyde dehydrogenase. During this reaction 2 NADH are produced. If large amounts of ethanol are present, then large amounts of NADH are produced, leading to a depletion of NAD+. Thus, the conversion of pyruvate to lactate is increased due to the associated regeneration of NAD+. Therefore, anion-gap metabolic acidosis (lactic acidosis) may ensue in ethanol poisoning.
The increased NADH/NAD+ ratio also can cause hypoglycemia in an (otherwise) fasting individual who has been drinking and is dependent on gluconeogenesis to maintain blood glucose levels. Alanine and lactate are major gluconeogenic precursors that enter gluconeogenesis as pyruvate. The high NADH/NAD+ ratio shifts the lactate dehydrogenase equilibrium to lactate, so that less pyruvate can be formed and, therefore, gluconeogenesis is impaired.
Isoencimas
A lactato deshidroxenase funcional é un homo ou heterotetrámero composto polas subunidades M e H codificads polos xenes LDHA do cromosoma 11 e LDHB do cromosoma 12, respectivamente. As posibles isoencimas son:
- LDH-1 (4H) — presente no corazón e glóbulos vermellos do sangue
- LDH-2 (3H1M) — no sistema reticuloendotelial
- LDH-3 (2H2M) — nos pulmóns
- LDH-4 (1H3M) — nos riles, placenta, e páncreas
- LDH-5 (4M) — no fígado e músculo esquelético[2]
O principal isoencimas presentes no fígado e músculo esquelético é o M4, que ten catro subunidades M (M do inglés muscle), mentres que o isoencima H4 é o principal na maioría das especies no corazón, con 4 subunidades H (H de heart). As outras variantes conteñen as dúas subunidades en diferentes proporcións, polo que son heterotetrámeras.
Xeralmente a LDH-2 é a forma predominante no soro sanguíneo. Un nivel de LDH-1 máis alto que o de LDH-2 (un "patrón invertido" con respecto ao normal) suxire infarto de miocardio (as lesións cardíacas liberan a LDH cardíaca LDH-1, no torrente sanguíneo).
Xenética en humanos
The M and H subunits are encoded by two different genes:
- The M subunit is encoded by LDHA, located on chromosome 11p15.4 (Online 'Mendelian Inheritance in Man' (OMIM) 150000)
- The H subunit is encoded by LDHB, located on chromosome 12p12.2-p12.1 (Online 'Mendelian Inheritance in Man' (OMIM) 150100)
- A third isoform, LDHC or LDHX, is expressed only in the testis (Online 'Mendelian Inheritance in Man' (OMIM) 150150); its gene is likely a duplicate of LDHA and is also located on the eleventh chromosome (11p15.5-p15.3)
Mutations of the M subunit have been linked to the rare disease exertional myoglobinuria (see OMIM article), and mutations of the H subunit have been described but do not appear to lead to disease.
Uso médico
LDH is a protein that normally appears throughout the body in small amounts. Many cancers can raise LDH levels, so LDH may be used as a tumor marker, but at the same time, it is not useful in identifying a specific kind of cancer. Measuring LDH levels can be helpful in monitoring treatment for cancer. Noncancerous conditions that can raise LDH levels include heart failure, hypothyroidism, anemia, and lung or liver disease.[3]
Tissue breakdown releases LDH, and therefore LDH can be measured as a surrogate for tissue breakdown, e.g. hemolysis. Other disorders indicated by elevated LDH include cancer, meningitis, encephalitis, acute pancreatitis, and HIV. LDH is measured by the lactate dehydrogenase (LDH) test (also known as the LDH test or Lactic acid dehydrogenase test). Comparison of the measured LDH values with the normal range help guide diagnosis.[4]
Hemólise
In medicine, LDH is often used as a marker of tissue breakdown as LDH is abundant in red blood cells and can function as a marker for hemolysis. A blood sample that has been handled incorrectly can show false-positively high levels of LDH due to erythrocyte damage.
It can also be used as a marker of myocardial infarction. Following a myocardial infarction, levels of LDH peak at 3–4 days and remain elevated for up to 10 days. In this way, elevated levels of LDH (where the level of LDH1 is higher than that of LDH2) can be useful for determining whether a patient has had a myocardial infarction if they come to doctors several days after an episode of chest pain.
Tissue turnover
Other uses are assessment of tissue breakdown in general; this is possible when there are no other indicators of hemolysis. It is used to follow-up cancer (especially lymphoma) patients, as cancer cells have a high rate of turnover with destroyed cells leading to an elevated LDH activity.
Exudados e transudados
Measuring LDH in fluid aspirated from a pleural effusion (or pericardial effusion) can help in the distinction between exudates (actively secreted fluid, e.g. due to inflammation) or transudates (passively secreted fluid, due to a high hydrostatic pressure or a low oncotic pressure). The usual criterion is that a ratio of fluid LDH versus upper limit of normal serum LDH of more than 0.6[5] or Modelo:Fraction[6] indicates an exudate, while a ratio of less indicates a transudate. Different laboratories have different values for the upper limit of serum LDH, but examples include 200[7] and 300[7] IU/L.[8] In empyema, the LDH levels, in general, will exceed 1000 IU/L.
Meninxite e encefalite
High levels of lactate dehydrogenase in cerebrospinal fluid are often associated with bacterial meningitis.[9] In the case of viral meningitis, high LDH, in general, indicates the presence of encephalitis and poor prognosis.
VIH
LDH is often measured in HIV patients as a non-specific marker for pneumonia due to Pneumocystis jiroveci (PCP). Elevated LDH in the setting of upper respiratory symptoms in an HIV patient suggests, but is not diagnostic for, PCP. However, in HIV-positive patients with respiratory symptoms, a very high LDH level (>600 IU/L) indicated histoplasmosis (9.33 more likely) in a study of 120 PCP and 30 histoplasmosis patients.[10]
Disxerminoma
Elevated LDH is often the first clinical sign of a rare malignant cell tumor called a dysgerminoma. Not all dysgerminomas produce LDH, and this is often a non-specific finding.
Procariotas
A cap-membrane-binding domain is found in prokaryotic lactate dehydrogenase. This consists of a large seven-stranded antiparallel beta-sheet flanked on both sides by alpha-helices. It allows for membrane association.[11]
Notas
- ↑ Selwood T, Jaffe EK (2012). "Dynamic dissociating homo-oligomers and the control of protein function". Arch. Biochem. Biophys. 519 (2): 131–43. PMC 3298769. PMID 22182754. doi:10.1016/j.abb.2011.11.020. Parámetro descoñecido
|month=ignorado (Axuda) - ↑ Van Eerd, J. P. F. M.; Kreutzer, E. K. J. (1996). Klinische Chemie voor Analisten deel 2. pp. 138–139. ISBN 978-90-313-2003-5.
- ↑ Stanford Cancer Center. "Cancer Diagnosis - Understanding Cancer". Understanding Cancer. Stanford Medicine.
- ↑ "Lactate dehydrogenase test: MedlinePlus Medical Encyclopedia". MedlinePlus. U.S. National Library of Medicine.
- ↑ Heffner JE, Brown LK, Barbieri CA (1997). "Diagnostic value of tests that discriminate between exudative and transudative pleural effusions. Primary Study Investigators". Chest 111 (4): 970–80. PMID 9106577. doi:10.1378/chest.111.4.970. Parámetro descoñecido
|month=ignorado (Axuda) - ↑ Light RW, Macgregor MI, Luchsinger PC, Ball WC (1972). "Pleural effusions: the diagnostic separation of transudates and exudates". Ann. Intern. Med. 77 (4): 507–13. PMID 4642731. doi:10.7326/0003-4819-77-4-507. Parámetro descoñecido
|month=ignorado (Axuda) - ↑ 7,0 7,1 Joseph J, Badrinath P, Basran GS, Sahn SA (2001). "Is the pleural fluid transudate or exudate? A revisit of the diagnostic criteria". Thorax 56 (11): 867–70. PMC 1745948. PMID 11641512. doi:10.1136/thorax.56.11.867. Parámetro descoñecido
|month=ignorado (Axuda) - ↑ Joseph J, Badrinath P, Basran GS, Sahn SA (2002). "Is albumin gradient or fluid to serum albumin ratio better than the pleural fluid lactate dehydroginase in the diagnostic of separation of pleural effusion?". BMC Pulm Med 2: 1. PMC 101409. PMID 11914151. doi:10.1186/1471-2466-2-1. Parámetro descoñecido
|month=ignorado (Axuda) - ↑ Stein JH (1998). Internal Medicine. Elsevier Health Sciences. pp. 1408–. ISBN 978-0-8151-8698-4. Consultado o 12 August 2013.
- ↑ Butt AA, Michaels S, Greer D, Clark R, Kissinger P, Martin DH (2002). "Serum LDH level as a clue to the diagnosis of histoplasmosis". AIDS Read 12 (7): 317–21. PMID 12161854. Parámetro descoñecido
|month=ignorado (Axuda) - ↑ Dym O, Pratt EA, Ho C, Eisenberg D (2000). "The crystal structure of D-lactate dehydrogenase, a peripheral membrane respiratory enzyme". Proc. Natl. Acad. Sci. U.S.A. 97 (17): 9413–8. PMC 16878. PMID 10944213. doi:10.1073/pnas.97.17.9413. Parámetro descoñecido
|month=ignorado (Axuda)
Véxase tamén
Outros artigos
Este artigo incorpora contidos en dominio público de Pfam e InterPro IPR015409
